The OIF has been approved for the following funding awards through the Eugene Washington PCORI Engagement Awards program, an initiative of the Patient-Centered Outcomes Research Institute (PCORI):
Improving Patient-Centered Outcomes: Expanding Engagement of the Osteogenesis Imperfecta Community
Project Dates: 6/1/2019—5/31/2020
Project Summary: As individuals with OI age, a vast array of other collagen-based problems such as gastrointestinal disorders, loss of mobility, impaired dentition, cardiopulmonary deficits, and hearing loss are frequently dominant themes. Lacking is information reflecting the “patient’s voice” about research priorities, quality of life, patient-reported outcomes, clinical best practices, and the natural history of a broad range of clinical and treatment concerns. The absence of this information results in inefficient, fragmented, and perhaps inappropriate care with highly variable outcomes and unnecessary expense. Funding from the Patient-Centered Outcomes Research institute (PCORI) would be used to leverage the ongoing work of the OI Foundation (OIF). Our proposed two-year project has five specific aims; expand the OI stakeholder community focused on performing patient-centered outcomes research (PCOR); expand existing OIF communication and education strategies; establish and extend capacity among the OI community to participate in PCOR activities; develop an OI specific PCOR toolkit and extend the OI PCOR/CER approach and products to support other rare bone disease communities. Achieving these objectives will help improve patient care by engaging all stakeholders (patients, caregivers, clinicians, and researchers) in identifying and prioritizing current gaps in care and treatment.
To achieve these objectives, the project will engage two stakeholder groups: the OI community and the clinicians and researchers that serve them. A diverse PCORI Advisory Board will be recruited from these two stakeholder groups, as will Communications and Education Committees. Communication protocols will be devised that both inform and help to gather important feedback from the patient community. Educational programs describing the import of PCOR will be made a key component of all on-going OIF educational events. Participation from Rare Bone Disease Alliance members will be sought to support both the Board and the two committees.
At the completion of this project, we will have 1) created a community of stakeholders–patient/caregiver/clinicians/researchers — trained in and committed to engaging in PCOR, with specific attention to research topics that the OI community regards as high priority; 2) expanded existing OIF communication and education strategies to create lasting relationships with engaged audiences that ultimately support sustainable participation in PCOR and achieve measurable, usable results that enhance the care of the OI community; 3) established or extended capacity among OI patients, caregivers, clinicians, and researchers in the OI community to participate in PCOR and comparative effectiveness (CER) activities; 4) developed an OI-specific PCOR Toolkit that facilitates sustainable input from the community and promotes dissemination of evidence-based clinical care recommendations to the stakeholder and clinical care community; and 5) extended our OI PCOR/CER approach and products to support other rare bone disease communities.
COVID-19 Enhancement Award
Project Dates 7/1/2020—8/31/2021
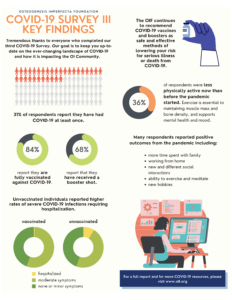
Project Summary: The enhancement to this project will complement the existing project by ensuring the osteogenesis imperfecta community and the clinicians who treat them are aware of the evolving effects of COVID-19 as they seek to build capacity for PCOR/CER. The Osteogenesis Imperfecta Foundation will convene a panel of experts who will serve as a COVID-19 Task Force. The project team will expand the focus of the PCOR training and toolkit they will create under the existing award to include COVID-19. This inclusion is expected to provide insight into priorities for osteogenesis imperfecta patients.
Leveraging Virtual Communication to Advance PCOR Adoption by the Rare Bone Disease Community
Project Dates 7/1/2021—6/30/2022
Project Summary: Rare Bone Diseases account for 5% of all birth defects and virtually all rare bone diseases cause significant physical disabilities. Patients and caregivers are frequently isolated and uncertain about best care practices. Using virtual technology allows patients and clinicians to connect and to gain knowledge of their rare bone disorder. The project Leveraging Virtual Communication to Advance PCOR Adoption by the Rare Bone Disease Community will work to leverage the relationships among members of the Rare Bone Disease Alliance to host virtual meetings with leaders and stakeholders as well as clinician/researchers who often care for several rare diseases. The Rare Bone Disease Alliance is a network of patient groups and professionals dedicated to improving the lives of people with various rare bone diseases. The aim of the project is to share the successful PCOR experience of the Osteogenesis Imperfecta Foundation with the rare bone disease community. The project will pay particular attention to reaching stakeholders from underrepresented communities. To achieve this goal, the project will convene a series of virtual meetings over one year that enhances collaboration among rare bone disease communities; increases underrepresented community participation in PCORI; engages with stakeholders who may be disadvantaged by the digital divide; initiates an engagement approach that fosters collaboration and partnerships; and develops a road map to implement and sustain robust stakeholder engagement built solely on virtual platforms. The project will convene a series of 3 large group virtual meetings that will introduce PCOR to the rare bone disease communities. In addition, the project will hold several focus group virtual meetings with patients and caregivers to ascertain the barriers to expert care from the patient perspective while identifying potential collaborative clinical research opportunities. The 13 Rare Bone Disease Alliance patient organizations are invited to make up the project’s Advisory Committee along with other stakeholders, including rare bone expert clinicians. The Osteogenesis Imperfecta Foundation serves as the lead patient organization supported by OI expert and orthopedic surgeon Dr. Laura Tosi and Duke University’s Dr. Bryce Reeve, from its Populations Sciences Department, as subcontractors.
Preparing Adults with Osteogenesis Imperfecta to Engage in Research on Access and Quality of Care for Their Rare Disease
Project Dates: 11/01/2021—10/31/2023
Project Summary: Most pediatric OI patients (up to age 18-22) are cared for at one of over 60 OI clinics located around the country. Only 5 clinics, however, care for OI patients who are considered of “adult” age. The lack of adult OI care in the US poses significant problems for transition to adult care and access to quality specialty care particularly for individuals living in geographically isolated areas.
To begin to address this problem, we propose to prepare the OI community to be able to compare different strategies for delivering high quality care for widely dispersed individuals with a complex disorder. We will accomplish this by first expanding the reach of our patient-centered outcomes research education efforts to include a diverse group of stakeholders from across the US. We will then use listening sessions, interviews, and surveys to explore how patient-centered outcomes research might identify opportunities for improving adult OI care. We will explore problems that young adults with OI have as they transition from pediatric to adult care, challenges those older adults with OI encounter as they seek primary and specialty care, difficulties that pediatric OI clinics have as they seek to successfully transition their patients to adult care, and barriers and facilitators that existing adult OI clinics encounter as they seek to provide appropriate and high-quality care to adult patients.
At the completion of this project, we will have significantly expanded the knowledge, competencies, and abilities of persons with OI and other stakeholders to be meaningful partners in future PCOR and CER studies. We will have significantly strengthened the skills of researchers to be better partners with patients and other stakeholders involved in PCOR/CER. We will pursue an aggressive dissemination program, specifically including publications in both the peer-reviewed literature and OI Foundation communications. Over the short term (0-2 years), the findings of this project will provide the foundation for developing future PCOR/CER projects which will identify optimal approaches for enhancing transition and adult care for individuals with OI over the long term (3+ years).

















 Patient Advocate Spotlight: Tracy Hart Fosters Connections to Help Patients with Osteogenesis Imperfecta
Patient Advocate Spotlight: Tracy Hart Fosters Connections to Help Patients with Osteogenesis Imperfecta





